Applications
Accelerate the development of cancer immunotherapies, at every stage
VivaCyte enables a diverse set of applications throughout the cell therapy development cycle

Discovery
- Comprehensive immune cell characterization
- Deep analysis of mechanism of action
- High-throughput screening of CRISPR-edited cells or TCR-T cells

Preclinical
- Immuno-monitoring of in vivo studies
- Advanced functional
screening of exhaustion
and persistence

Translational
- Immuno-monitoring based on potency of persistent cells
- Correlation of clinical outcome with potency assessment
- Development of biomarkers of enhanced anti-tumor activity
Process Dev
- High throughput process development and optimization
- Rapid and in-depth batch characterization
- Donor validation

Manufacturing
- Walk-away potency assays based on cytotoxicity
- Walk-away potency assays based on cytokine release
- Multiplex analysis of potency and phenotype
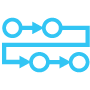
Automate cell-mediated cytotoxicity assays with a minimum amount of sample
Generate an entire dose-response curve per microfluidic channel, up to 16 dose-response curves per CC-Array plate, using only a few thousand effector cells per channel. Bulk cytotoxicity workflows available for suspension target cells.
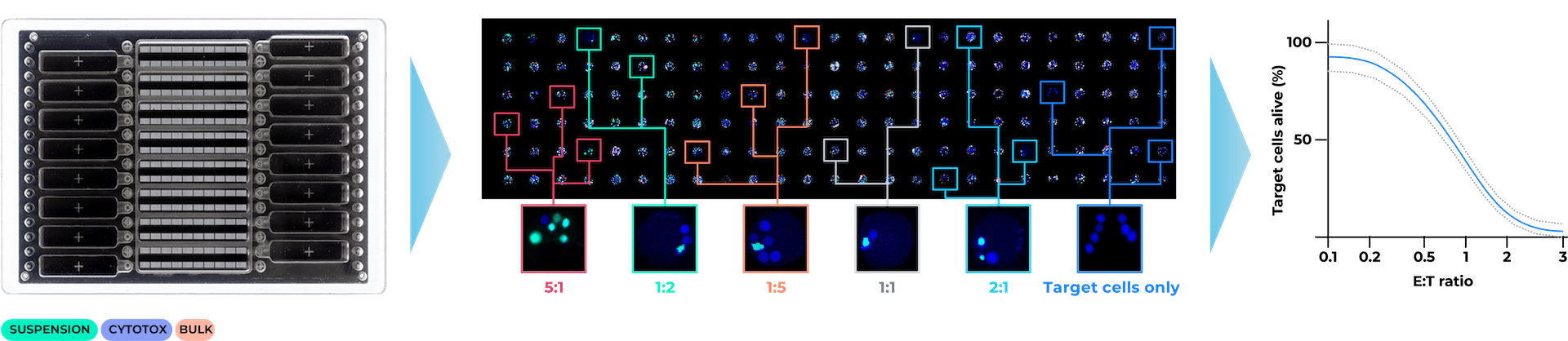
Unravel the heterogeneity of your cell therapy samples
Single-cell multiparametric analysis of cytotoxicity and phenotype enables unprecedented depth of analysis:
- Rapidly define frequency of killer and serial killer cells
- Measure potency of cell subsets, e.g. CD4 and CD8, in a single assay using just one microfluidic channel per sample
- Generate a unified potency score, highly reproducible across runs
Combined with parallel screening of multiple conditions in a single run, multiparametric analysis enables comprehensive testing of cell therapy candidates from numerous process options, against multiple target cells, to rapidly identify the most suitable candidates.
Find your supercharged cells
A single immune cell can be evaluated for immunophenotype and ability to kill two or more target cells within a time lapsed study. Leveraging this protocol, the CC-Array technology allows quantification of serial killing at a single-cell resolution in just 24 hours instead of weeks needed by conventional bulk assays. The manual tedious work required in re-challenge assays is replaced with a fully automated process requiring just 20 minutes hand-on time.

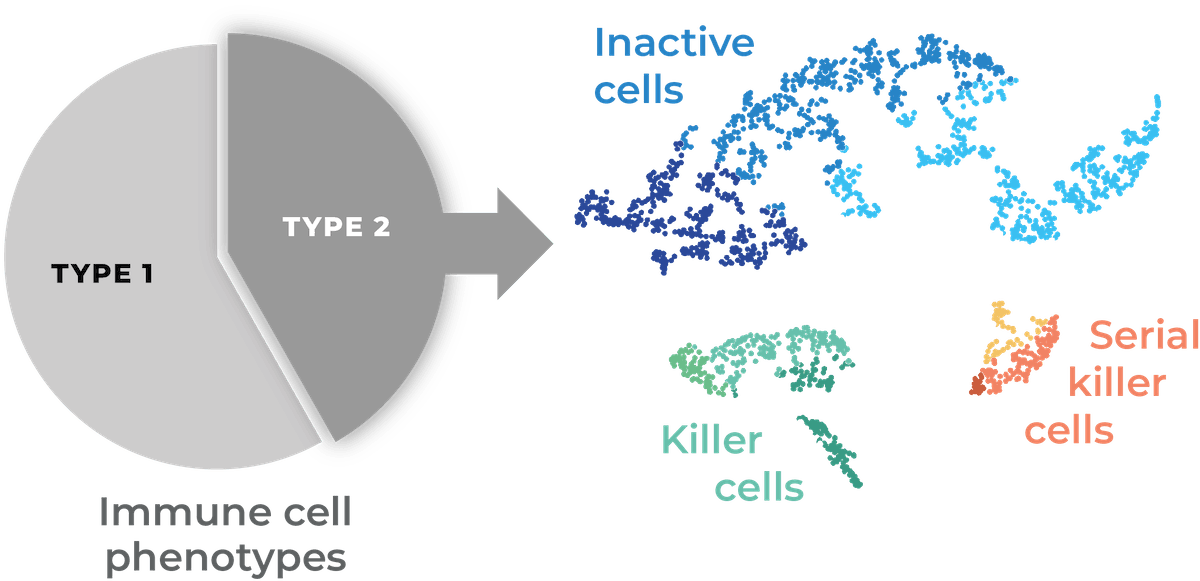
Comparability through a Unified Potency Score
A Unified Potency Score (UPS) is calculated from single-cell cytotoxicity data providing a single indicator for the potency of your cells.
Frequency of both killer and serial killer cells contribute to the UPS, for a comprehensive evaluation of the potency of your product.
Once an incubation time and target cell line are defined, the UPS (time, target) is a standardized score allowing direct comparison of multiple products or product candidates, irrespective of the source of your cells, the batch or cell therapy type.
Resources
TN-005_High throughput parallel screening of multiple Effector : Target (E:T) ratios for cell mediated cytotoxicity kinetic analysis using low sample volume
Learn how VivaCyte can generate multiple dose-response analyses in a single CC-Array plate, based on a cell-mediated cytotoxicity assay carried out at multiple E:T ratios
TECHNICAL NOTE | Document number: TN-005
TN-006_A novel method for comparing the potency of CAR-NK cells to support informed donor selection
Introducing a novel Unified Potency Score (UPS) to quantify the potency of cell therapy products with a single indicator, generated by single-cell killing and serial killing data
TECHNICAL NOTE | Document number: TN-006
TN-007_Characterization of NK cell donors for allogeneic cell therapy using single cell serial killing
Learn how a single-cell serial killing assay allows deep characterization of immune cell function and enables ranking of donors for allogeneic NK cell therapy
TECHNICAL NOTE | Document number: TN-007
TN-008_How single-cell multiparametric analysis enables in-depth understanding of functional heterogeneity and killer kinetics in CAR-T cell therapies
Learn how multiparametric analysis allows to define the potency of subpopulations of cells in a single assay through a combined single-cell analysis of cytotoxicity and phenotype carried out on each immune cell